Introduction
Sulfur is the fifth most common on Earth yet comprises a major portion of objectionable environmental effects. Much of the sulfur is contained in coal and crude oil deposits, which can commonly contain 1-2% sulfur by weight. If the sulfur is burned in a fuel stream, the resulting products include SOx, which form corrosive products when condensing in the presence of water vapor. Despite active efforts towards energy transition, fossil fuels will continue to dominate the energy demand in the foreseeable future. Thus, there is a need for novel adsorbents and catalysts to remove sulfur components more completely, and more efficiently. When sulfur is removed from fuel streams, this enables the furnaces to be designed and operated with additional heat recovery from the flue gas, without worry of acid gas condensation from the SOx.
This article examines a few key applications of sulfur removal where advancements in adsorbent and catalyst formulations lead to greater levels of sulfur removal, which are combined with process energy savings.
Sulfur Removal
Since 2014, GTC Vorro (GTCV) has been involved in the sulfur removal business offering its solutions for various applications such as well-head gas, fuel gas, refinery fuels desulfurization etc. Over the past decade, GTCV, in collaboration with its partners, has developed sulfur removing media and processes for different applications in the industry.
NanoSØRB™
NanoSØRB™ is a Ni/ZnO nanowire-based catalytic adsorbent, which removes sulfur from natural gas, syngas, fuel gas, LPG, gasoline, kerosene, and diesel. The technology operates at lower pressure, temperature, and hydrogen-to-oil ratio than other processes. The adsorbent is capable of removing H2S, COS, mercaptans, disulfides and thiophenes, and achieving TOTAL Sulfur specification of < 1 ppm or lower, if needed.
The spent media is regenerable using air for oxidizing the adsorbed sulfur into SO2. The regeneration gas can be routed to a Claus unit for final conversion into elemental sulfur. It is important to note that NanoSØRB™ is not conventional hydrodesulfurization (HDS). There is no H2S production during the desulfurization process in NanoSØRB™. Ni extracts the S from the sulfur contained hydrocarbon species and Zn converts the sulfur into ZnS. The fundamental advantage of using nanowires as opposed to nano-spheres, is that nanowires retain significantly high surface area throughout the operation and activation. In contrast, nanospheres are prone to sintering upon activation. Due to the ideal crystal surface in nanowires, the media displays high selectivity and reactivity. Hence, the operating parameters required for desulfurization are significantly less than conventional hydrodesulfurization.

Fig 1: Nanowires vs Nanospheres: Sintering
SweetTreat-O™
SweetTreat-O™ is a proprietary mixed metal oxide adsorbent, which was developed to have ultra-high H2S removal capacity. SweetTreat-O™ can achieve 110-120 wt% sulfur loading. The media has a highly porous structure, which results in low pressure drop across the system. Further, due to the usage of certain metals in the formulation, the adsorbent exhibits fast adsorption kinetics as opposed to slow kinetics in traditional iron-oxide adsorbents.
The adsorbent is deployed in a system of two identical fixed bed scavengers in “lead/lag” configuration, which allows maximum operation flexibility. A small amount of air is introduced in the system, to maintain a certain O2/H2S ratio via an air blower to provide oxygen for inline regeneration of the adsorbent. When the adsorbent in the lead scavenger approaches the end of life and needs to be replaced, this vessel can be taken offline for adsorbent changeout; and the lag scavenger will keep operating without any compromise on the product specification. The fundamental chemistry behind adsorption is as follows:
M-O + H2S → M-S + H2O (Adsorption)
M-S + 1/O2 → M-O + elemental S (Inline Regeneration)
[where M denotes the metal content in the media]
Due to the porosity of the media, elemental sulfur deposits inside the pores, while the gas is flowing on the outside of the pore across the active sites. Hence, the system does not experience high pressure drop as the sulfur is removed. Eventually, the elemental sulfur blocks the active sites leading to media becoming spent. The spent media is classified as Non-Hazardous Commercial Waste and can be disposed of at a landfill.
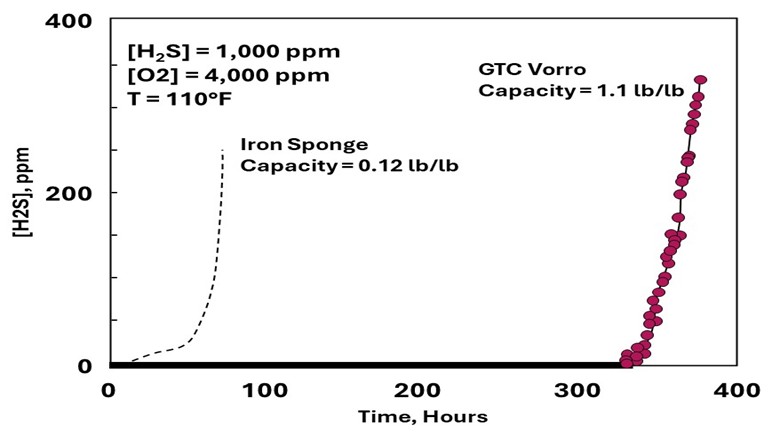
Fig 2: Media capacity comparison with iron sponge
Desulfurization Applications
This section outlines key applications of the media, wherein the operator can achieve Sulfur Removal, Energy Efficiency, and Decarbonization.
Fuel Gas Desulfurization
Fuel gas is a primary energy source in different industries, and depending on its origination, can contain different sulfur species.
- In the steel industry, when reducing gas is used for iron-ore reduction, a slipstream from the reducing gas reactor outlet is fed to onsite fired heater along with utility natural gas. The sulfur form is only H2
- In NGL gas processing, in particular propane and butane, molecular sieves are used for mercaptan removal; and fuel gas is used for molecular sieve regeneration. Post regeneration, fuel gas is sent to a fired heater to recover its energy value, along with utility natural gas. In such instances, the combined fuel gas typically contains mercaptans and H2
- In small- to mid-scale LNG plants, molecular sieves are sometimes used for CO2, H2S, mercaptan, and H2O removal. Typically fuel gas is used for molecular sieve regeneration, and post regeneration, it can be routed back to the source pipeline or used as fuel in fired heaters. In such scenarios, sulfur species present in fuel gas can be H2S, mercaptans, and disulfides.
- Refinery fuel gases contain the most varied mix of sulfur species such as COS, H2S, mercaptan, or disulfides. The nature depends on the upstream units of hydroprocessing, FCC, or Delayed Coker. Generally, the TOTAL Sulfur content in refinery fuel gas is in the range 20-100 ppmv, controlled by an amine unit that operates to maintain an overall site limit for sulfur emission. Further, refinery fuel gas invariably contains H2.
It is important to understand the nature of sulfur species to implement the right media solution for fuel gas desulfurization, which can lead to utility natural gas savings along with partial decarbonization. Referring to Fig 3 any sulfur species present in the fuel gas will get converted to SO2 after combustion in the furnace. In general, 2-4% of this SO2 gets converted into SO3 due to excess O2 in the furnace. Condensation of this SO3 leads to a highly corrosive acid environment. Hence, during operations, it is critical to run the air preheater such that flue gas outlet temperature does not drop below the acid dewpoint. Another factor to consider while operating is to avoid the area of high sulfuric acid corrosion rate at 100-150 deg C. In other words, hypothetically even if the acid dew point of flue gas is 120 deg C, the flue gas air preheater outlet should still be kept above 160 deg C to prevent any condensation happening within the preheater tubes due to transient anomalies in upstream operations or feedstock sulfur content, leading to higher than anticipated Total S in the fuel gas.
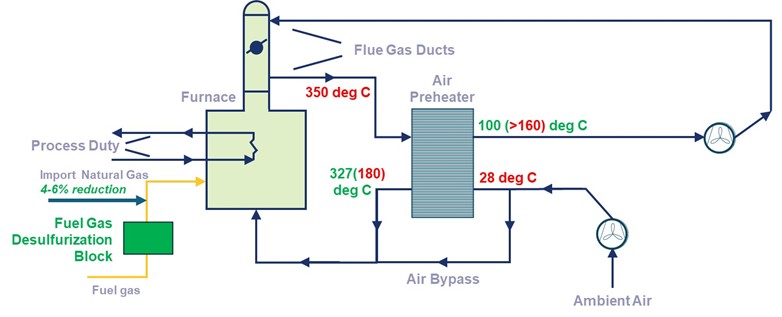
Fig 3: Fuel Gas Desulfurization implementation in fired heaters. RED denominates typical operation while GREEN indicates optimized operation post desulfurization.
The fuel gas desulfurization block, offered by GTCV, can reliably and consistently reduce the TOTAL sulfur in fuel gas below 1 ppmv. This will lead to acid dewpoint reduction in the range 75-85 deg C, enabling the operator to lower the air preheater flue gas outlet temperature to < 100 deg C. This can lead to 4-6% savings in import natural gas. The entire system’s payback period, depending on the natural gas price, will be less than one year, if proper flue gas heat recovery is used. If the existing air preheater is limited by area or duty to recuperate the additional heat, then GTCV can offer a comprehensive package of additional heat pipe-based air preheater along with fuel gas desulfurization. The additional air preheater will be located downstream of the existing air preheater.
As mentioned earlier, the nature of sulfur species dictates the media deployed for fuel gas desulfurization. If only H2S is present, then SweetTreat-OTM is the chosen approach. If fuel gas contains other sulfur species like mercaptans, disulfides etc, then the media of choice is NanoSØRB™. For refinery fuel gas, since it inherently contains H2, no external H2 is needed. However, if the fuel gas doesn’t contain H2, like in the case of mol sieve regen fuel gas, external H2 is needed to be mixed with feed prior to entering the NanoSØRB™ block. Due to the significant high surface area of the NanoSØRB™ catalytic adsorbent, fuel gas preheating and hydrogen requirements are significantly less as compared to conventional hydrodesulfurization step. It is important to note that usage of non-regenerative adsorbents like copper oxide, for removing various sulfur species, is technically non-viable due to the presence of H2 in refinery fuel gas stream. H2 will reduce the CuO to copper rendering the system non-functional in operation.
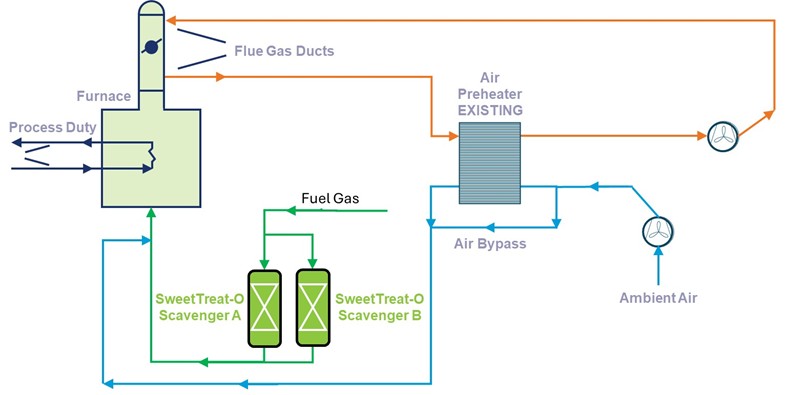
Fig 4: Fuel Gas Desulfurization when only H2S is present.
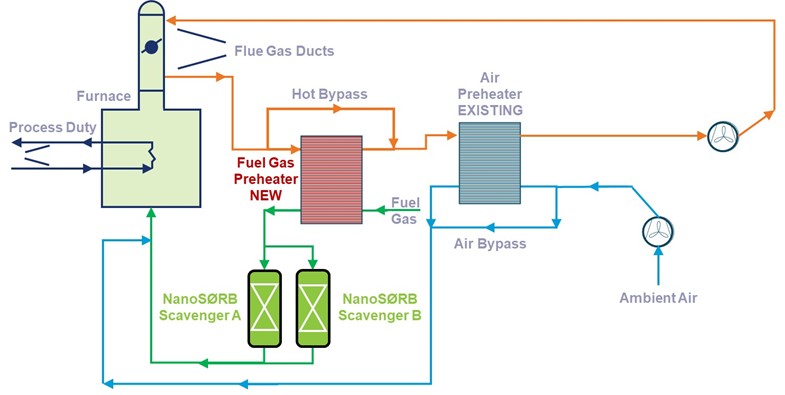
Fig 5: Fuel Gas Desulfurization when different sulfur species, H2S, mercaptans, disulfides, etc., are present.
Coal/Petcoke Syngas Sulfur Removal
During petcoke/coal gasification, most of the sulfur contained in the feedstock, is converted into H2S. However, 3-10% gets converted into COS; and, depending on the initial feedstock sulfur content and downstream usage of syngas, this can create problems in meeting the desired specifications or require additional, complex unit operations to meet the required specifications.
- For chemical synthesis applications which require syngas with less than 1 ppmv sulfur, physical solvent processes such as RectisolTM, offered by Linde and Air Liquide, and SelexolTM, offered by DOW, are sometimes used for sulfur removal.. These processes operate at chilled conditions (Rectisol operates at -40°C)1. However, if the syngas pressure is low, the physical solvent processes might require compression, which can lead to significant power consumption.
- For power generation or fuel gas applications, which allow higher sulfur levels (approximately 10 to 30 ppmv sulfur), chemical solvent processes such as MDEA are normally used to selectively reject some of the CO However, these solvents do not capture COS. The COS removal is often accomplished by hydrolysis via the reaction:
COS + H2O ↔ H2S + CO2
This is an equilibrium reaction and generally requires two steps with intermediate H2S removal. After 1st stage hydrolysis, an amine pre-absorber is used to remove the bulk H2S followed by 2nd stage hydrolysis and again an amine absorption. A common regeneration section for both amine absorbers is implemented as depicted in Fig 6. The reason for amine pre-wash is to further promote COS hydrolysis due to the equilibrium nature of reaction.
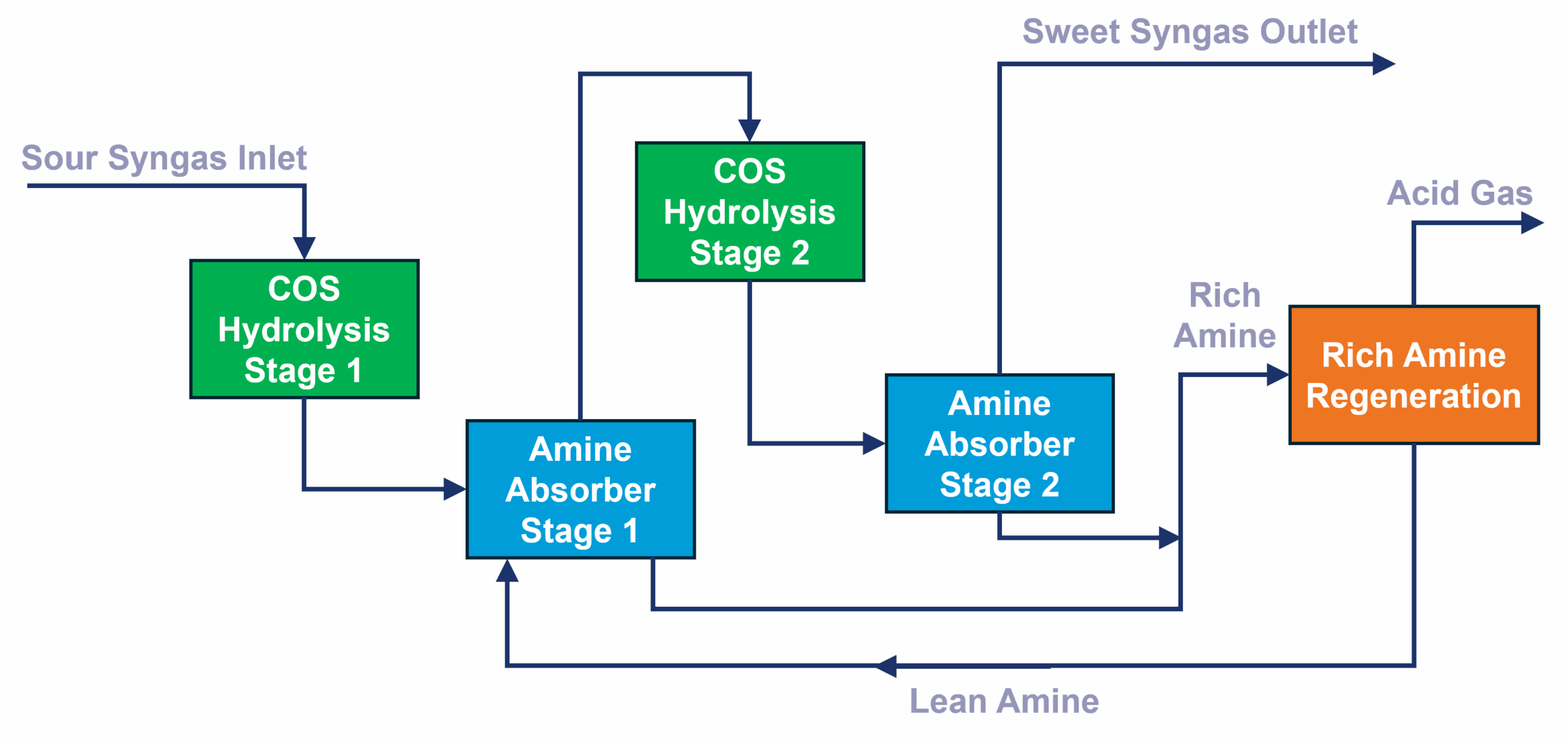
Fig 6: 2 stage COS hydrolysis and MDEA section.
GTCV offers single-stage proprietary amine followed by NanoSØRB™, which can remove COS, H2S and other sulfur species if present, to <1 ppmv levels. The proprietary amine is formulated to have high selectivity towards H2S as compared to CO2 leading to high H2S concentration in the acid gas. This is required to maintain efficient operation of the Claus unit as well as ensure stable operation of the furnace. COS is not absorbed by the proprietary amine, and an optimized H2S level is allowed to slip through the amine contactor. The remaining H2S and COS are then removed to < 1 ppmv levels across the NanoSØRB™ block. See Fig 7 below:

Fig 7: GTCV offering of proprietary amine followed by NanoSØRB™
The process significantly reduces the CAPEX and OPEX requirements as compared to any physical or chemical solvent-based process, and achieves the intended downstream sulfur specification of syngas for any usage.
Conclusion
Due to advancements made in sulfur removal media such as NanoSØRB™ and SweetTreat-OTM, operators can enjoy the benefits of sulfur removal along with energy efficiency. Fuel gas and Syngas desulfurization are two applications addressed in this article. However, the usage of these media transcends these two applications and can be applied for LPG, FCC gasoline, diesel and other sulfur removal applications as well.
References
https://www.netl.doe.gov/research/coal/energy-systems/gasification/gasifipedia/cleanup

